General hydrochemistry
The physical and chemical characteristics of groundwater samples collected from the Quaternary, Upper Miocene and Lower Miocene aquifers are summarised in Table 5, which presents minimum, maximum, mean and standard deviation values for each parameter alongside the World Health Organisation51 drinking water guidelines. Examination of these data reveals distinct hydrochemical signatures across the three aquifer systems.
The pH values of groundwater from the Lower Miocene aquifer ranged from 6.0 to 7.7, with a mean of 6.61. In the Upper Miocene aquifer, pH varied between 6.4 and 7.8, averaging 6.84, while Quaternary aquifer samples exhibited pH values from 6.3 to 7.4 with a mean of 6.75. These ranges indicate slightly acidic to slightly alkaline conditions across all aquifers, potentially attributable to anthropogenic influences or natural biogeochemical processes. Electrical conductivity, a proxy for total dissolved ion concentration, demonstrated considerable variability both within and between aquifers. The Lower Miocene aquifer exhibited EC values from 1150 to 62850 μS/cm, with a mean of 12451 μS/cm. Upper Miocene aquifer samples ranged from 777 to 22380 μS/cm, averaging 7601 μS/cm, while Quaternary aquifer samples varied between 335 and 29460 μS/cm, with a mean of 9261 μS/cm. This wide range reflects substantial heterogeneity in groundwater mineralisation, likely resulting from variable degrees of aquifer interaction with saline sources. Total dissolved solids concentrations corroborated the EC patterns. Lower Miocene aquifer samples contained TDS between 717.31 and 48449 mg/l, mean 8112 mg/l. Upper Miocene aquifer values ranged from 505.58 to 12884 mg/l, mean 4076 mg/l, and Quaternary aquifer samples exhibited TDS from 174 to 16452 mg/l, mean 5377 mg/l. Comparison with WHO51 drinking water standards indicates that the majority of samples exceed permissible limits, with only 2, 7 and 8% of samples from the Lower Miocene, Upper Miocene and Quaternary aquifers respectively falling within acceptable ranges for potable use.
The major ion chemistry reveals sodium as the dominant cation, with mean concentrations of 2158.17 mg/l in the Lower Miocene, 893 mg/l in the Upper Miocene and 1192 mg/l in the Quaternary aquifer. Chloride similarly dominates the anion composition, with mean values of 3902.12 mg/l, 1821.51 mg/l and 2524 mg/l respectively. This sodium-chloride dominance suggests significant seawater influence or halite dissolution as primary salinity sources. Calcium and magnesium concentrations are also elevated, with mean calcium values of 384.31 249.6 and 232.5 mg/l across the three aquifers, and corresponding magnesium means of 462.21, 722.78 and 318.9 mg/l.
The seawater fraction calculations, based on chloride concentrations as a conservative tracer52, indicate substantial marine influence. Lower Miocene aquifer samples exhibited seawater fractions up to 123.76%, with a mean of 17.91%, suggesting some samples represent pure seawater or brines exceeding seawater concentration through evaporation. Upper Miocene aquifer samples reached 24.3% seawater fraction, mean 8.29%, while Quaternary aquifer samples attained 43.99%, mean 11.54%. These values confirm variable but widespread seawater intrusion affecting all three aquifer systems. Saturation indices calculated for relevant mineral phases provide insight into geochemical evolution. All aquifers are supersaturated with respect to dolomite, barite and gibbsite, indicating thermodynamic conditions favouring precipitation of these minerals. Conversely, undersaturation with respect to calcite, anhydrite, gypsum and halite suggests these phases remain undersaturated and continue to dissolve where present, contributing to ongoing mineralisation. Total hardness calculations following Hem (1985 classify the majority of samples as very hard. In the Quaternary aquifer, 7% of samples are hard and the remainder very hard; all Upper Miocene aquifer samples are very hard; and Lower Miocene aquifer samples, containing the highest seawater fractions, are uniformly very hard. These hardness characteristics have significant implications for both domestic and agricultural use.
The water quality index, computed from electrical conductivity, magnesium hazard, sodium adsorption ratio, sodium percentage, Kelly index and permeability index, provides an integrated assessment of suitability for various applications. Results indicate that 91% of Lower Miocene aquifer samples, 89% of Upper Miocene samples and 67% of Quaternary samples are categorised as unsuitable for drinking purposes, requiring appropriate treatment prior to use. A smaller proportion (5, 11 and 10% respectively) is classified as poor quality but potentially suitable for industrial and irrigation applications with appropriate management. Very poor quality, restricted to possible irrigation use only, characterises 3% of Lower Miocene and 8% of Quaternary aquifer samples.
Ionic relations and the origin of solutes
The stoichiometric relationships among dissolved ions provide diagnostic information regarding the sources of salinity and the geochemical processes operating within the aquifer systems. These relationships, examined through binary ionic plots, enable discrimination between seawater intrusion, mineral dissolution and ion exchange mechanisms. The relationship between sodium and chloride concentrations (Fig. 5a) exhibits a strong positive correlation (R2 = 0.773), indicating a common source or coupled behaviour. Samples plot both on and above the 1:1 halite dissolution line, with Na/Cl molar ratios ranging from 0.529 to 2.519 in the Lower Miocene aquifer, 0.245 to 1.285 in the Upper Miocene aquifer and 0.058 to 1.124 in the Quaternary aquifer. The elevated chloride concentrations relative to sodium in many samples suggest seawater influence, while sodium excess in others indicates additional sodium sources such as silicate weathering or cation exchange reactions. The relationship between sulphate and calcium (Fig. 5b) informs assessment of gypsum and anhydrite dissolution. Where sulphate exceeds calcium on an equivalent basis, the implication is that calcium has been removed from solution through precipitation of calcite or participation in ion exchange reactions. Examination of this relationship across the dataset reveals variable stoichiometry, with some samples exhibiting sulphate excess consistent with calcium removal processes. The (calcium + magnesium) versus bicarbonate relationship (Fig. 5c) demonstrates clustering of most samples near the 1:1 line, suggesting that dissolution of atmospheric carbon dioxide in fresh water contributes to the acquisition of these ions. However, in many samples, particularly those from deeper wells with elevated salinity, calcium and magnesium exceed bicarbonate concentrations, indicating additional sources of these divalent cations beyond carbonate dissolution.

Ionic relationships for collected groundwater samples.
Processes controlling groundwater salinization
The Gibbs diagram53 provides a conceptual framework for distinguishing the dominant natural mechanisms governing groundwater chemistry: atmospheric precipitation, rock weathering and evaporation-crystallisation. Plotting the collected groundwater samples on Gibbs diagrams (Fig. 6) reveals that evaporation and rock-water interaction constitute the primary processes controlling solute acquisition across the study area. Samples from all three aquifers plot predominantly within the evaporation dominance field and the transition zone toward rock weathering dominance. This distribution indicates that evaporative concentration, whether occurring prior to recharge or within the aquifer system itself, plays a substantial role in elevating salinities. The absence of samples plotting within the precipitation dominance field confirms that atmospheric inputs are not the primary control on groundwater chemistry in this region.

Gibbs plot for collected water samples.
The Chadha diagram54, a modified version of the Piper diagram, enables classification of groundwaters according to their hydrochemical facies. This rectangular diagram is divided into eight sub-fields representing distinct water types based on the differences between alkaline earths and alkali metals on the abscissa and between weak acidic anions and strong acidic anions on the ordinate. As illustrated in (Fig. 7), the majority of groundwater samples from the study area fall within Field 3, characterised by alkali metals exceeding alkaline earths, with sodium dominance among cations and chloride dominance among anions. This hydrochemical facies is typical of waters influenced by seawater intrusion or halite dissolution, consistent with the elevated sodium and chloride concentrations documented in Table 2. A secondary group of samples occupies Field 2, where calcium and magnesium are the dominant cations, indicating zones where carbonate dissolution exerts greater influence on water chemistry.

Chadha diagram for collected water samples.
Sources of salinity and its associated process on water chemistry
Cation exchange reactions are intimately associated with salinisation processes in coastal aquifers, as documented extensively in the literature55,56 These reactions modify the relative proportions of cations in solution as fresh water and salt water interact, with enrichment or depletion of specific cations providing diagnostic indicators of exchange direction and intensity. The seawater fraction calculations presented in Section “General hydrochemistry”, based on chloride as a conservative tracer52,57, quantify the extent of marine influence in each sample. The substantial seawater fractions observed, particularly in the Lower Miocene aquifer where values reach 123.76%, confirm that seawater intrusion constitutes a primary salinisation mechanism. Values exceeding 100% indicate samples that have undergone evaporative concentration beyond simple seawater mixing, consistent with the evaporation dominance indicated by Gibbs diagrams. Saturation indices computed for relevant mineral phases provide additional constraints on geochemical evolution. The observed supersaturation with respect to dolomite, barite and gibbsite, coupled with undersaturation for calcite, anhydrite, gypsum and halite, indicates that precipitation of the former minerals may occur while the latter phases remain capable of dissolution where present. This combination of saturated and undersaturated phases reflects the complex, multi-source evolution of groundwater salinity in these aquifer systems. The water quality categorisation presented in Table 6 synthesises the hydrochemical data into an integrated assessment of suitability for various applications. The predominance of unsuitable classifications across all aquifers (91% Lower Miocene, 89% Upper Miocene, 67% Quaternary) reflects the combined effects of seawater intrusion, evaporative concentration and mineral dissolution processes documented throughout this analysis. The small proportions of poor and very poor-quality water, potentially suitable for irrigation with appropriate management, are restricted to samples exhibiting lesser degrees of saline influence.
Aquifer-specific isotopic characteristics and implications for site suitability
Stable environmental isotopes (δ1⁸O and δD) serve as intrinsic tracers within the hydrological cycle, with their relative abundances modified by physical processes including evaporation, condensation and mixing. These variations provide diagnostic information regarding groundwater origin, recharge mechanisms and interactions between different water bodies. In the context of site suitability assessment for green hydrogen production, the isotopic composition of groundwater informs the availability and sustainability of freshwater resources required for electrolysis, as well as the extent of seawater intrusion that may compromise water quality. Stable environmental isotopes (δ1⁸O and δD) are intrinsic constituents of water molecules, with their relative abundances altered by physical and mass-dependent processes during the hydrological cycle. These variations reflect the origin, movement, and interactions of water within hydrogeological systems. The isotopic composition of groundwater is influenced by meteorological conditions (e.g., temperature, humidity), source-specific signatures (rainfall, surface water, seawater), and secondary processes (evaporation, mixing). In the study area, potential recharge sources exhibit distinct isotopic fingerprints (Table 7):
The isotopic composition of groundwater exhibits distinct signatures across the three aquifer systems, reflecting differences in recharge history, hydraulic connectivity and dominant geochemical processes. These variations carry direct implications for the availability and sustainability of water resources for green hydrogen production.
Quaternary aquifer
Groundwater from the Quaternary aquifer exhibits mean δ1⁸O of − 4.16‰ and mean δD of − 18.65‰ (Table 8), values substantially depleted relative to modern Mediterranean precipitation (δ1⁸O: − 3.24‰; δD: − 11.25‰). This depletion indicates that a significant component of recharge occurred under cooler and/or wetter climatic conditions than those prevailing today. The observed range (δ1⁸O: − 6.60 to 0.93‰; δD: − 33.50 to 5.84‰) reflects mixing between paleowater and recent recharge, with the most enriched samples (δ1⁸O approaching 0.93‰) indicating evaporative modification in shallow zones or mixing with saline sources. The δ1⁸O-δD relationship (δD = 4.731 δ1⁸O + 1.0357; R2 = 0.9048) yields a slope considerably lower than the Global Meteoric Water Line (GMWL: δD = 8 δ1⁸O + 10;60), demonstrating that evaporation has modified the isotopic composition prior to or during recharge. The strong linear correlation (R2 = 0.9048) indicates a consistent evaporative regime across the aquifer, with samples representing varying degrees of enrichment along a common evaporation trajectory. The positive correlation between δ1⁸O and TDS (Fig. 8b) is consistent with evaporative concentration, wherein water loss through evaporation simultaneously enriches residual water in heavy isotopes and increases dissolved solute concentrations. Samples approaching the seawater end-member (δ1⁸O = + 1.76‰; TDS = 35,000 mg/l) delineate zones where marine intrusion contributes to groundwater salinisation.
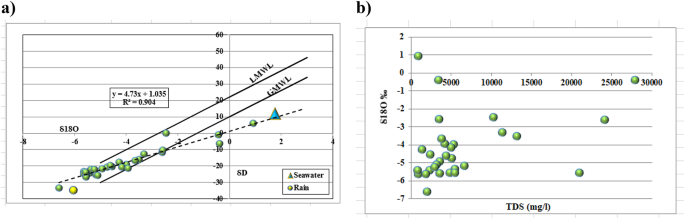
Binary relationship for quaternary aquifer between; (a) δ1⁸O and δD and (b) δ18O (‰) and TDS (mg/l).
Miocene aquifers
The Miocene aquifers display more depleted isotopic signatures than the Quaternary aquifer, with mean δ1⁸O of − 4.73‰ and mean δD of − 24.00‰ for both Upper and Lower units (Table 8). The most depleted samples (δ1⁸O: − 10.35‰; δD: − 80.29‰) approach the composition of Nubian Sandstone paleowater (δ1⁸O: − 10.0‰; δD: − 80.0‰), suggesting that deep circulation from the regional aquifer system contributes to Miocene groundwater in some areas. The wide range in δD values (standard deviation 23.40‰) relative to δ1⁸O (3.48‰) reflects variable mixing between isotopically distinct end-members.
The δ1⁸O-δD relationships for Miocene samples, Fig. 9a) define two distinct regression lines: Upper Miocene (δD = 5.0387 δ1⁸O + 1.1504; R2 = 0.8507) and Lower Miocene (δD = 4.5369 δ1⁸O − 2.0142; R2 = 0.8198). Both slopes are lower than the GMWL, indicating evaporative influence, though the Upper Miocene samples exhibit a slightly higher slope, suggesting less evaporative modification or greater contribution from depleted paleowater. The Lower Miocene regression line exhibits a negative intercept (− 2.0142), consistent with mixing between evaporated modern water and a depleted paleowater component. The elevated R2 values demonstrate that mixing between end-members, rather than random variation, controls the observed isotopic distribution. The relationship between δ1⁸O and TDS for Miocene samples (Fig. 9b) differs markedly from the Quaternary aquifer. Despite a wide TDS range (approximately 2000 to > 40,000 mg/l), δ1⁸O values remain relatively constrained between − 6‰ and − 2‰. This pattern indicates that salinity acquisition in the Miocene aquifers occurs primarily through water–rock interactions—including dissolution of evaporite minerals, cation exchange and mixing with connate brines—rather than evaporative concentration. The absence of a strong δ1⁸O-TDS correlation suggests that saline samples have acquired their dissolved solids through subsurface processes that do not fractionate oxygen isotopes.

Binary relationship for Miocene aquifer (lower and upper) between; (a) δ1⁸O and δD and (b) δ18O (‰) and TDS (mg/l).
Synthesis and site suitability implications
The isotopic evidence yields three principal findings relevant to site suitability assessment for green hydrogen production:
First, the Quaternary aquifer receives measurable modern recharge, as evidenced by isotopic values extending toward the local meteoric water line and the positive δ⁸O-TDS correlation indicative of ongoing evaporative processes. However, elevated salinities in coastal zones and isotopic evidence for seawater mixing indicate that groundwater quality is compromised near the Mediterranean shoreline. Sites underlain by the Quaternary aquifer at sufficient distance from the coast may access water suitable for electrolysis following appropriate treatment, though the evaporative signature in shallow groundwater implies vulnerability to seasonal and climatic variations in recharge quality.
Second, the Miocene aquifers contain predominantly paleowater with limited modern recharge, as indicated by depleted isotopic signatures and the absence of an evaporation-TDS relationship. The most depleted samples approach the composition of Nubian paleowater, suggesting that deep, fossil groundwater contributes to these aquifers. While water quality is highly variable, the deep, confined nature of the Lower Miocene aquifer may provide a more stable and protected resource, albeit one that is effectively non-renewable on human timescales. Any abstraction for hydrogen production would require careful assessment of sustainable yield and consideration of the non-renewable nature of the resource.
Third, isotopic evidence for seawater intrusion in coastal portions of both aquifer systems delineates zones where groundwater is unsuitable for electrolysis without substantial desalination. The mixing relationships quantified through δ1⁸O-δD regression lines provide a basis for mapping the extent of marine influence and identifying areas where fresh groundwater remains protected. Samples exhibiting δ1⁸O values above −2‰ and elevated TDS are clearly influenced by seawater and should be avoided for hydrogen production without desalination infrastructure.
These hydrochemical and isotopic constraints, when integrated with the economic and environmental criteria discussed previously, inform the identification of sites where adequate water resources of suitable quality can be secured for long-term hydrogen production operations. The evidence suggests that optimal sites will be located inland, away from the coastal seawater intrusion zone, and underlain by aquifers with sufficient freshwater reserves to support sustained abstraction.



