The common laboratory practice of using sacrificial agents to boost photocatalytic hydrogen evolution is holding back the development of viable solar‑hydrogen technologies. That’s according to scientists who are calling on the community to clearly distinguish between sacrificial agent-assisted systems and true overall water splitting when reporting their research in the literature.
Photocatalytic water splitting is an alluring prospect, with its promise of generating hydrogen from nothing more than sunlight and water attracting researchers seeking alternatives to fossil fuels.
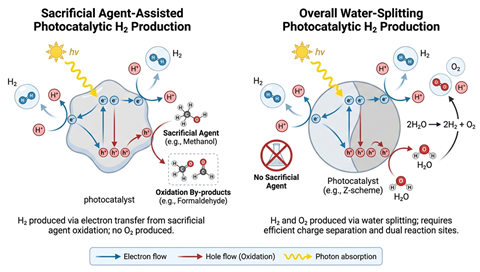
The process begins when sunlight hits a semiconductor photocatalyst, generating an electron–hole pair. The holes oxidise water to produce oxygen and protons. And hydrogen is produced when protons are reduced by the electrons. This simultaneous evolution of hydrogen and oxygen from water is referred to as photocatalytic overall water splitting.
Despite its apparent simplicity, photocatalytic water splitting has struggled to mature into a practical route to green hydrogen. Catalysts must drive oxidation and reduction reactions simultaneously while remaining stable and charge-balanced, and even then, the process faces kinetic challenges because the oxygen evolution reaction is unfavourable.
To sidestep these hurdles, large swathes of photocatalytic hydrogen evolution research rely on sacrificial agents. However, some researchers, including Mohammad Rahman at Chengdu University in China, warn that the sustained reliance on these approaches is hindering progress toward sustainable, scalable solar hydrogen production via overall water splitting.
At odds with sustainability
While Rahman acknowledges the value of sacrificial agents for mechanistic studies and catalyst development, he and colleagues point to a mismatch between sacrificial hydrogen evolution and the goal of producing hydrogen solely from water and renewable energy. ‘[Sacrificial] systems are experimentally simpler and can deliver higher apparent performance. However… much of the reported progress does not directly translate into practical hydrogen production technologies,’ says Rahman.
Sacrificial reagents, such as alcohols and amines, suppress unwanted electron–hole recombination and provide an alternative, more favourable oxidation pathway. Instead of oxygen, the sacrificial reagent is oxidised to a byproduct, bypassing the oxygen evolution reaction. This boosts hydrogen yields and apparent efficiency, and allows the use of photocatalysts tailored specifically for hydrogen evolution.
Takashi Hisatomi, an expert in photocatalytic water splitting at Shinshu University in Japan, says ‘the widespread use of sacrificial systems is often not a matter of preference or policy, but rather an inevitable choice due to the extreme difficulty of the overall water splitting reaction itself.’
Rahman and colleagues’ argument rests on four premises: that genuinely green hydrogen must come from the direct, renewable splitting of water; that bypassing the oxygen evolution reaction introduces non‑renewable consumables and generates waste products; that sacrificial systems distort performance metrics and distract from the development of true overall water‑splitting catalysts; and that the environmental and economic costs of scaling sacrificial agents are incompatible with the promise of green hydrogen.
These premises emphasise that photocatalytic water splitting should aim for a stoichiometric reaction, simultaneously generating hydrogen and oxygen. And while the cost of lab-scale sacrificial agents is manageable, industrial reactions would require enormous quantities of expensive consumables, many of which are derived from fossil fuels. By circumventing the oxygen evolution reaction, sacrificial systems instead oxidise these reagents to waste products, creating additional disposal or processing burdens. Together, these factors render sacrificial approaches financially and environmentally unsustainable at scale.
Refocussing the field
Although sacrificial systems have advanced the understanding and design of photocatalytic hydrogen evolution, Rahman and colleagues note that the inflated reaction rates they deliver do not automatically carry over to overall water splitting. They say sacrificial systems should instead be assessed on mechanistic and diagnostic grounds.
They also call for the development of standardised protocols and accredited benchmarks to enable clearer comparisons between systems. Hisatomi agrees that these proposals offer meaningful guidance for the field, adding that ‘moving away from evaluation based on sacrificial reagents is an essential and inevitable step toward the practical application of green hydrogen production.’
However, Hisatomi goes on to highlight a ‘fundamental bottleneck that no material system capable of achieving practical solar-to-hydrogen energy-conversion efficiency has yet been established.’ This, he says, is a barrier to such proposals moving from academic theory to reality. ‘Only when a robust photocatalytic system that requires no specialised conditions is realised, will the proposed certification and standardisation gain practical efficacy … the top priority must be the establishment of a reliable, highly-active standard photocatalyst system.’
Rahman and colleagues argue that action is needed to bridge the gap between sacrificial systems and scalable overall water splitting. They call for experimental frameworks, evaluation metrics and research incentives that are aligned with stoichiometric water splitting, and stress that clearly distinguishing between simplified laboratory systems and sustainability‑driven studies is essential. Such differentiation, they say, would help preserve insights gained from sacrificial systems while accelerating progress toward credible photocatalysts that operate solely on water and sunlight. ‘The next step is not to replace these approaches, but to build on them more consciously,’ adds Rahman. ‘A small shift in how we frame results, design experiments, and define success could have a large cumulative impact.’



